Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124

Allow me to begin by bringing to your attention the fact that, in an electric vehicle the battery is very important for it holds electrical energy required to fuel a motor.
To better understand its composition, here is an overview of its architecture:
Formation of an Electric Car Battery
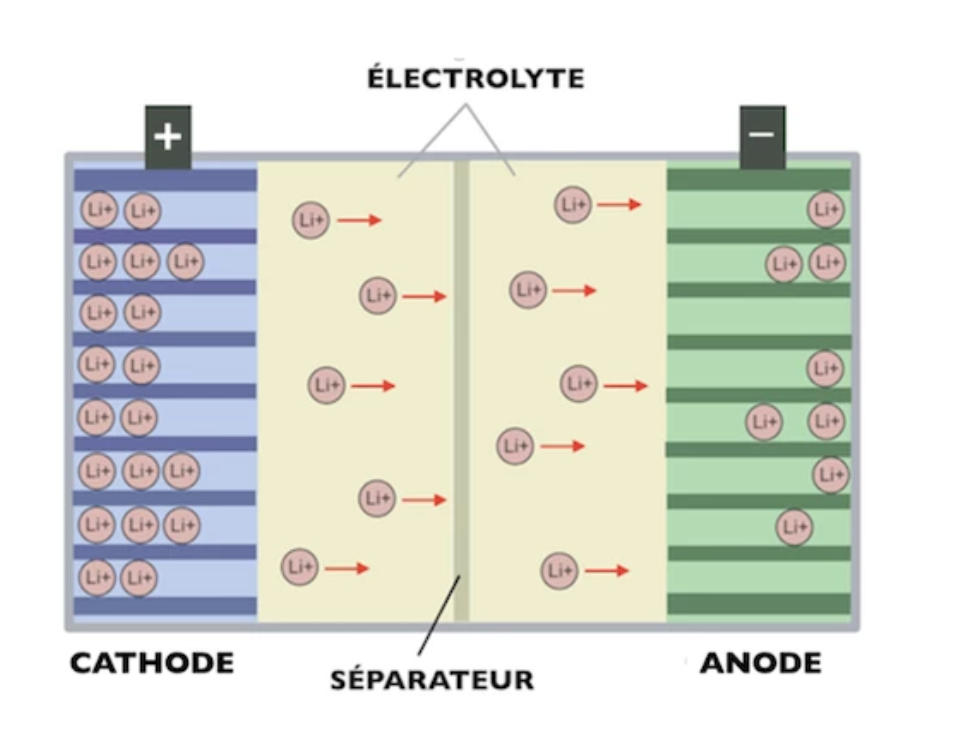
A simplified diagram shows us more about the composition of an electric car battery, which consists of several key elements: an anode and a cathode, a salt bridge, and an electrolyte, and an insulating material known as a separator. Let’s disaggregate these components.
Electrodes
The electrodes are the vital parts of an electric car battery. There are two types of electrodes: the anode and the cathode.
How They Work:
Composition of Anodes and Cathodes:
Both are normally constructed from active materials which are capable of storing electrical energy. Lithium is common but it is not the only material that can be utilized.
The Electrolyte
It is a liquid (or solid) material in which the ions can travel across between the electrodes which include the anode and the cathode. It is generally represented by an organic solvent containing lithium salts.
How It Works:
The Separator
Purpose of the Separator:
The separator keeps anode and cathode from making physical contact, to avoid short circuit and hence failure of the battery. Yet the wall which surrounds this space is not completely airtight.
The separator prevents electrons from flowing straight across from an anode to the cathode. Electrons are compelled to flow through the external contacts of the battery in the course of a circuit.
Why is lithium used in batteries of electric cars?
Lithium is commonly used in electric car batteries because it possesses unique property of high efficiency with power storage and discharge. Its attributes make it suitable for high power, light and long-range battery systems, all characteristics of modern EVs. Despite such advancements, still, the search for other materials to enhance the efficiency of batteries and their cost and recyclability goes on.
It is much easier to grasp what goes on inside an electric car battery, and why these components are so crucial to the future of greener automobiles, when one understands the general layout of an electric car battery as presented above.